Abstract
Research Article
Effects of Vochysia haenkeana extract on the neuromuscular blockade induced by Bothrops jararaca venom on chick biventer cervicis preparation in vitro
Yoko Oshima-Franco*, Fernanda Dias da Silva, Natália Tribuiani, Isadora Caruso Fontana Oliveira, Regina Yuri Hashimoto Miura, Rafael S Floriano, Márcio Galdino dos Santos and Sandro Rostelato-Ferreira
Published: 08 August, 2017 | Volume 1 - Issue 1 | Pages: 052-058
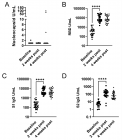
Vochysia haenkeana extract (Vh-E) was assessed against the neuromuscular blockade induced by Bothrops jararaca venom on chick biventer cervicis (BC) preparation. Pre- and post-venom incubation treatments (Pre-vit and Post-vit) were analysed here. Contractures ACh (110 µM) and KCl (20 mM) were evoked before and after addition of venom without stimulation. Vh-E (600 µg/mL) under Pre-vit was more efficient to neutralize the neuromuscular blockade by venom (40 µg/mL) [72.5±4.6% (venom) vs. 45.2±14% (Vh-E) of blockade, p<0.05, n=4]. Vh-E (600 µg/mL) did not cause significant changes under Post-vit [72.5±4.6% (venom) vs. 63.4±8.2% (Vh-E) of blockade, n=4]. The Pre-vit inhibited the blockade of the contracture to ACh (106±17% of response; n=4) while the Post-vit was able to attenuate the effect of the venom on this contracture (55±5% of response; n=4); related to those contractures to KCl both of treatments with Vh-E attenuated the blocker effect of the venom (62.5±7.7% and 55±5% of response for Pre-vit and Post-vit, respectively; n=4). In conclusion, Vh-E neutralizes partially the neuromuscular blockade in Pre-vit, an effect that can be related to preserved function of “extrinsic” post-synaptic receptors, by measured contractures in response to ACh. The myotoxicity of the venom was significantly reduced by Vh-E in both, Pre-vit and Post-vit, by measured contractures in response to KCl.
Read Full Article HTML DOI: 10.29328/journal.jpsp.1001006 Cite this Article Read Full Article PDF
References
- WHO World Health Organization. Snake antivenoms. Fact sheet No 337. Reviewed February 2015. Ref.: https://goo.gl/ZeqZyB
- Oliveira LS, Muzitano MF, Coutinho MAS, Giany Oliveira de Melo, Sônia Soares Costa. Plantas medicinais como recurso terapêutico em comunidade do entorno da reserva biológica do Tinguá, RJ, Brasil - metabólitos secundários e aspectos farmacológicos. Revista Científica Internacional. 2011; 4: 54-74. Ref.: https://goo.gl/s6pc2z
- Indriunas A, Aoyama EM. Plantas empregadas em acidentes ofídicos de Systema Materiae Medicae Vegetabilis Brasiliensis de Martius. II Simpósio sobre a biodiversidade da Mata Atlântica. 2013; 189-194.
- Moura VM, Mourão RHV, Santos MCD. Acidentes ofídicos na região norte do Brasil e o uso de espécies vegetais como tratamento alternativo e complementar à soroterapia. Scientia Amazonia. 2015; 4: 73-84.
- Werner AC, Ferraz MC, Yoshida EH, Tribuiani N, Gautuz JA, et al. The facilitatory effect of Casearia sylvestris Sw. (guaçatonga) fractions on the contractile activity of mammalian and avian neuromuscular apparatus. Current Pharmaceutical Biotechnology. 2015; 16: 468-481. Ref.: https://goo.gl/kt9SuX
- Costa RB, Contini AZ, Melo ESP. Reproductive system of Anadenanthera peregrina and Vochysia haenkiana in a fragment of "Cerrado forest" from Chapada dos Guimarães - MT, Brazil. Ciência Rural. 2003; 33: 305-310. Ref.: https://goo.gl/62N3UP
- Cezari EJ. Plantas medicinais: atividade antitumoral do extrato bruto de sete plantas do cerrado e o uso por povos tradicionais. [Dissertação]. Universidade Federal do Tocantins. Palmas. 2010.
- Collaço RdeC, Cogo JC, Rodrigues-Simioni L, Rocha T, Oshima-Franco Y, et al. Protection by Mikania laevigata (guaco) extract against the toxicity of Philodryas olfersii snake venom. Toxicon. 2012; 60: 614-622. Ref.: https://goo.gl/hCZnda
- Ginsborg BL, Warriner J. The isolated chick biventer cervicis nerve-muscle preparation. British Journal of Pharmacology and Chemotherapy. 1960; 15. 410-411. Ref.: https://goo.gl/TYB3pU
- Harvey AL, and Marshall IG. Skeletal muscle. In: Sturkie’s avian physiology (Wittow GC ed.), 5th ed, San Diego London, Boston, New York, Sydney, Tokyo, Toronto: Academic Press. 2000; 8: 123-139.
- Santos MG, Lolis SF, Dal Belo CA. Levantamentos etnobotânicos realizados em duas comunidades de remanescentes de negros da região do Jalapão, Estado do Tocantins. In: Pires AICS, Oliveira R. (orgs.) Sociabilidades negras. Comunidades remanescentes, escravidão e cultura. Belo Horizonte: Daliana Ltda. 2006.
- Lima PC, dos Santos MG, Calabrese KS, et al. Evaluation of the leishmanicidal activity of plant species of the Brazilian savana. Revista de Patologia Tropical. 2015; 44: 45-55.
- Amaral CFS, Silva OA, Godoy P, Miranda D. Renal cortical necrosis following Bothrops jararaca and B. jararacussu snakebite. Toxicon. 1985; 23: 877-885. Ref.: https://goo.gl/UFJfJV
- Oshima-Franco Y, Alves CMV, Andréo Filho N, Gerenutti M, Cintra ACO, et al. Neutralization of the neuromuscular activity of bothropstoxin-I, a myotoxin from Bothrops jararacussu snake venom, by a hydroalcoholic extract of Casearia sylvestris Sw. (guaçatonga). Journal of Venomous Animals and Toxins Including Tropical Diseases. 2005; 11: 465-478. Ref.: https://goo.gl/PbvGDW
- Cintra-Francischinelli M, Silva MG, Andréo-Filho N, N. Andréo-Filho, M. Gerenutti, et al. Antibothropic action of Casearia sylvestris Sw. (Flacourtiaceae) extracts. Phytotherapy Research. 2008; 22: 784-790. Ref.: https://goo.gl/zm1pMg
- Farrapo NM, Silva GAA, Costa KN, Magali Silva G, José Cogo C, et al. Inhibition of Bothrops jararacussu venom activities by Plathymenia reticulata Benth extracts. Journal of Venom Research. 2011; 2: 52-58. : https://goo.gl/JTyJ83
- Melo RS, Farrapo NM, Rocha DS, et al. Antiophidian mechanisms of medicinal plants. In: Keller RB (Ed.), Flavonoids: Biosynthesis, Biological Effects and Dietary Sources. Nova Science Publishers. 2009; 8: 249-262. Ref.: https://goo.gl/dv1BPe
- Nazato VS, Rubem-Mauro L, Vieira NAG, Rocha-Junior Ddos S, Silva MG, et al. In vitro antiophidian properties of Dipteryx alata Vogel bark extracts. Molecules. 2010; 15: 5956-5970. Ref.: https://goo.gl/s3jTwq
- Rosa LJR, Amaral Filho J, Silva MG, Magali Glauzer Silva, José Carlos Cogo, et al. The inhibitory effect of Camellia sinensis extracts against the neuromuscular blockade of Crotalus durissus terrificus venom. Journal of Venom Research. 2010; 1: 1-7. Ref.: https://goo.gl/doM89N
- Oshima-Franco Y, Rosa LJR, Silva GAA, et al. Antibothropic action of Camellia sinensis extract against the neuromuscular blockade by Bothrops jararacussu snake venom and its main toxin, bothropstoxin-I. In: Luca Gallelli (Ed). Pharmacology. Intech. 2012.
- Dal Belo DA, Lucho AP, Vinadé L, Rocha L, Seibert França H, et al. In vitro antiophidian mechanisms of Hypericum brasiliense Choisy standardized extract: quercetin-dependent neuroprotection. Biomed Research International. 2013; 2013: 943520. Ref.: https://goo.gl/XR5ybj
- Tribuiani N, Silva AM, Ferraz MC, Silva MG, Bentes AP, et al. Vellozia flavicans Mart. ex Schult. hydroalcoholic extract inhibits the neuromuscular blockade induced by Bothrops jararacussu venom. BMC Complementary and Alternative Medicine. 2014; 14: 48. Ref.: https://goo.gl/9bqdMt
- Camargo TM, Nazato VS, Cogo JC, Cogo JC, Groppo FC, et al. Bothrops jararacussu venom-induced neuromuscular blockade inhibited by Casearia gossypiosperma Briquet hydroalcoholic extract. Journal of Venomous Animals and Toxins including Tropical Diseases. 2010; 16: 432-441. Ref.: https://goo.gl/WB6rCi
- Soares-Silva JO, Oliveira JL, Cogo JC, et al. Pharmacological evaluation of hexane fraction of Casearia gossypiosperma Briquet: antivenom potentiality. Journal of Life Sciences. 2014; 8: 306-315.
- Ferreira-Rodrigues SC, Rodrigues CM, Dos Santos MG, Jean Antonio Abraham Gautuz, Magali Glauzer Silva, et al. Anti-inflammatory and antibothropic properties of Jatropha elliptica, a plant from Brazilian cerrado biome. Advanced Pharmaceutical Bulletin. 2016; 6: 573-579. Ref.: https://goo.gl/zcgZjZ
- Vatampour H. Effects of black scorpion Androctonus crasicuda venom on striated muscle preparation in vitro. Iranian Journal of Pharmaceutical Research. 2003; 2: 17-22. Ref.: https://goo.gl/7rhfxf
- Zamunér SR, da Cruz-Höfling MA, Corrado AP, Stephen Hyslop, LéaRodrigues S. Comparison of the neurotoxic and myotoxic effects of Brazilian Bothrops venoms and their neutralization by commercial antivenom. Toxicon. 2007; 44: 259-271. Ref.: https://goo.gl/jpbkKE
- Chang CC, Tang SS. Differentiation between intrinsic and extrinsic acetylcholine receptors of the chick biventer cervicis muscle. Naunyn-Schmiedeberg's Archives of Pharmacology. 1974; 282: 379-388. Ref.: https://goo.gl/iyqpSe
- Ginsborg BL. Some properties of avian skeletal muscle fibres with multiple neuromuscular junctions. Journal of Physiology. 1960; 154: 581-598. Ref.: https://goo.gl/kdFsSW
- Nelson BR, Wu F, Liu Y, Douglas M. Anderson, John McAnally, et al. Skeletal muscle-specific t-tubule protein stac3 mediates voltage-induced Ca2+ release and contractility. Proceedings of the National Academy of Sciences of the United States of America. 2013; 110: 11881-11886. Ref.: https://goo.gl/RzheLo
- Harvey AL, Barfaraz A, Thomson E, Faiz A, Preston S, et al. Screening of snake venoms for neurotoxic and myotoxic effects using simple in vitro preparations from rodents and chicks. Toxicon. 1994; 32: 257-265. Ref.: https://goo.gl/JzGNqc
- Barfaraz A, and Harvey AL. The use of the chick biventer cervicis preparation to assess the protective activity of six international reference antivenoms on the neuromuscular effects of snake venoms in vitro. Toxicon. 1994; 32: 267-272. Ref.: https://goo.gl/xo8hQC
Figures:

Figure 1

Figure 2
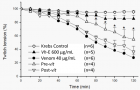
Figure 3

Figure 4

Figure 5
Similar Articles
-
Impact of Calcium Phosphate Nanoparticles on Rice PlantHrishikesh Upadhyaya*,Lutfa Begum,Bishal Dey,P K Nath,S K Panda. Impact of Calcium Phosphate Nanoparticles on Rice Plant. . 2017 doi: 10.29328/journal.jpsp.1001001; 1: 001-010
-
The Effects of Pharmacological Carbonic Anhydrase Suppression on Defence Responses of Potato Leaves To Phytophthora InfestansMagdalena Arasimowicz-Jelonek*,Jolanta Floryszak-Wieczorek. The Effects of Pharmacological Carbonic Anhydrase Suppression on Defence Responses of Potato Leaves To Phytophthora Infestans. . 2017 doi: 10.29328/journal.jpsp.1001002; 1: 011-025
-
Phytochemical content of leaf and stem of Marsilea quadrifolia (L.)Rajangam Udayakumar*,Karikalan Gopalakrishnan. Phytochemical content of leaf and stem of Marsilea quadrifolia (L.). . 2017 doi: 10.29328/journal.jpsp.1001003; 1: 026-037
-
Antagonistic features displayed by Plant Growth Promoting Rhizobacteria (PGPR): A ReviewMohsin Tariq*,Muhammad Noman,Temoor Ahmed,Amir Hameed,Natasha Manzoor,Marriam Zafar. Antagonistic features displayed by Plant Growth Promoting Rhizobacteria (PGPR): A Review . . 2017 doi: 10.29328/journal.jpsp.1001004; 1: 038-043
-
Wild-type Agrobacterium rhizogenes-mediated gene transfer in plants: Agrobacterium virulence and selection of transformantsShu Wei*,Muhammad Abdullah,Ferdinand L Shamalla,Mohammad M Rana. Wild-type Agrobacterium rhizogenes-mediated gene transfer in plants: Agrobacterium virulence and selection of transformants. . 2017 doi: 10.29328/journal.jpsp.1001005; 1: 044-051
-
Effects of Vochysia haenkeana extract on the neuromuscular blockade induced by Bothrops jararaca venom on chick biventer cervicis preparation in vitroYoko Oshima-Franco*,Fernanda Dias da Silva,Natália Tribuiani,Isadora Caruso Fontana Oliveira,Regina Yuri Hashimoto Miura,Rafael S Floriano,Márcio Galdino dos Santos,Sandro Rostelato-Ferreira. Effects of Vochysia haenkeana extract on the neuromuscular blockade induced by Bothrops jararaca venom on chick biventer cervicis preparation in vitro. . 2017 doi: 10.29328/journal.jpsp.1001006; 1: 052-058
-
HBV: Genomic Structure, HBVsAg Isolation and innovative Virotherapy Initiation in the Middle EastAboul-Ata E Aboul-Ata*,Essam M Janahi,I M El-Kalamawy,Kathleen Hefferon,Amal Mahmoud. HBV: Genomic Structure, HBVsAg Isolation and innovative Virotherapy Initiation in the Middle East . . 2017 doi: 10.29328/journal.jpsp.1001007; 1: 059-061
-
Physiological impact of Zinc nanoparticle on germination of rice (Oryza sativa L) seedUpadhyaya H*,Roy H,Shome S,Tewari S,Bhattacharya MK,Panda SK. Physiological impact of Zinc nanoparticle on germination of rice (Oryza sativa L) seed . . 2017 doi: 10.29328/journal.jpsp.1001008; 1: 062-070
-
Effects of Site Factors on the Clonal Growth of Phyllostachys bambusoides f. shouzhu YiXiaohong Gan*,Lijuan Chen,Cuibin Tang,Xia Zhang. Effects of Site Factors on the Clonal Growth of Phyllostachys bambusoides f. shouzhu Yi. . 2017 doi: 10.29328/journal.jpsp.1001009; 1: 071-079
-
Evaluation of genetic diversity in germplasm of paprika (Capsicum spp.) using random amplified polymorphic DNA (RAPD) markersRueda-Puente EO*,Renganathan P,Ruíz-Alvarado C,Hernández-Montiel LG,Prasath Duraisamy. Evaluation of genetic diversity in germplasm of paprika (Capsicum spp.) using random amplified polymorphic DNA (RAPD) markers. . 2017 doi: 10.29328/journal.jpsp.1001010; 1: 080-086
Recently Viewed
-
COPD and low plasma vitamin D levels: Correlation or causality?Luca Gallelli*, Erika Cione,Stefania Zampogna, Gino Scalone. COPD and low plasma vitamin D levels: Correlation or causality? . J Pulmonol Respir Res. 2018: doi: 10.29328/journal.jprr.1001008; 2: 011-012
-
Environmental Factors Affecting the Concentration of DNA in Blood and Saliva Stains: A ReviewDivya Khorwal*, GK Mathur, Umema Ahmed, SS Daga. Environmental Factors Affecting the Concentration of DNA in Blood and Saliva Stains: A Review. J Forensic Sci Res. 2024: doi: 10.29328/journal.jfsr.1001057; 8: 009-015
-
Markov Chains of Molecular Processes of Biochemical MaterialsOrchidea Maria Lecian*. Markov Chains of Molecular Processes of Biochemical Materials. Int J Phys Res Appl. 2024: doi: 10.29328/journal.ijpra.1001076; 7: 001-005
-
Generation of Curved Spacetime in Quantum FieldSarfraj Khan*. Generation of Curved Spacetime in Quantum Field. Int J Phys Res Appl. 2024: doi: 10.29328/journal.ijpra.1001077; 7: 006-009
-
Optimizing Milk Safety: Applying Nuclear Techniques in X-ray Fluorescence Spectroscopy for Heavy Metal Quantification in Powdered Milk Consumed in SenegalPapa Macoumba Faye*, Djicknack Dione, Oumar Ndiaye, Moussa Hamady SY, Nogaye Ndiaye, Alassane Traore, Ababacar Sadikhe Ndao. Optimizing Milk Safety: Applying Nuclear Techniques in X-ray Fluorescence Spectroscopy for Heavy Metal Quantification in Powdered Milk Consumed in Senegal. Int J Phys Res Appl. 2024: doi: 10.29328/journal.ijpra.1001078; 7: 010-015
Most Viewed
-
Evaluation of Biostimulants Based on Recovered Protein Hydrolysates from Animal By-products as Plant Growth EnhancersH Pérez-Aguilar*, M Lacruz-Asaro, F Arán-Ais. Evaluation of Biostimulants Based on Recovered Protein Hydrolysates from Animal By-products as Plant Growth Enhancers. J Plant Sci Phytopathol. 2023 doi: 10.29328/journal.jpsp.1001104; 7: 042-047
-
Sinonasal Myxoma Extending into the Orbit in a 4-Year Old: A Case PresentationJulian A Purrinos*, Ramzi Younis. Sinonasal Myxoma Extending into the Orbit in a 4-Year Old: A Case Presentation. Arch Case Rep. 2024 doi: 10.29328/journal.acr.1001099; 8: 075-077
-
Feasibility study of magnetic sensing for detecting single-neuron action potentialsDenis Tonini,Kai Wu,Renata Saha,Jian-Ping Wang*. Feasibility study of magnetic sensing for detecting single-neuron action potentials. Ann Biomed Sci Eng. 2022 doi: 10.29328/journal.abse.1001018; 6: 019-029
-
Pediatric Dysgerminoma: Unveiling a Rare Ovarian TumorFaten Limaiem*, Khalil Saffar, Ahmed Halouani. Pediatric Dysgerminoma: Unveiling a Rare Ovarian Tumor. Arch Case Rep. 2024 doi: 10.29328/journal.acr.1001087; 8: 010-013
-
Physical activity can change the physiological and psychological circumstances during COVID-19 pandemic: A narrative reviewKhashayar Maroufi*. Physical activity can change the physiological and psychological circumstances during COVID-19 pandemic: A narrative review. J Sports Med Ther. 2021 doi: 10.29328/journal.jsmt.1001051; 6: 001-007

HSPI: We're glad you're here. Please click "create a new Query" if you are a new visitor to our website and need further information from us.
If you are already a member of our network and need to keep track of any developments regarding a question you have already submitted, click "take me to my Query."