Abstract
Review Article
Role of HECT ubiquitin protein ligases in Arabidopsis thaliana
Ying Miao*, Wei Lan and Weibo Ma
Published: 20 March, 2018 | Volume 2 - Issue 1 | Pages: 020-030
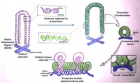
Ubiquitination is a kind of posttranslational modification of proteins in eukaryotes, and it plays an important role in the growth and development of organisms. The ubiquitination of proteins is a cascade enzymatic reaction involving three enzymes. The homologous to E6-AP carboxy terminus ubiquitin-protein ligases (HECT E3s) family is an important ubiquitin-protein ligases family. The family all have a HECT domain of approximately 350 amino acids in the C-terminus. However, studies on plant HECT E3s, such as structural features, prediction of HECT domain function, and their regulatory mechanisms, are very limited. In this paper, Arabidopsis thaliana HECT family genes were analyzed, including gene structure and functional domains and its limited known functions in protein degradation, gene transcription regulation, epigenetically regulation or other functions, finally speculate their roles in plant morphologies, aging or responsive to environmental stress.
Read Full Article HTML DOI: 10.29328/journal.jpsp.1001016 Cite this Article Read Full Article PDF
Keywords:
Arabidopsis thaliana; HECT E3 ubiquitin ligases; Ubiquitination; Biological function
References
- Goldberg AL. Protein degradation and protection against misfolded or damaged proteins. Nature. 2003; 426: 895-899. Ref.: https://goo.gl/v2K12U
- Luo H, Wong J, Wong B. Protein degradation systems in viral myocarditis leading to dilated cardiomyopathy. Cardiovasc Res. 2010; 85: 347-356. Ref.: https://goo.gl/WCVSpv
- Levine B, Mizushima N, Virgin HW. Autophagy in immunity and inflammation. Nature. 2011; 469: 323-335. Ref.: https://goo.gl/4t9rKm
- Pickart CM, Eddins MJ. Ubiquitin: structures, functions, mechanisms. Biochim Biophys Acta. 2004; 1695: 55-72. Ref.: https://goo.gl/iyJYyF
- Luzio JP, Pryor PR, Bright NA. Lysosomes: fusion and function. Nat Rev Mol Cell Biol.2007; 8: 622-632. Ref.: https://goo.gl/4XEDgR
- Husnjak K, Dikic I. Ubiquitin-binding proteins: decoders of ubiquitin-mediated cellular functions. Annu Rev Biochem. 2012; 81: 291-322. Ref.: https://goo.gl/qB7d7Z
- Kerscher O, Felberbaum R, Hochstrasser M. Modification of proteins by ubiquitin and ubiquitin-like proteins. Annu Rev Cell Dev Biol. 2006; 22: 159-180. Ref.: https://goo.gl/YJL8Hc
- Rotin D, Kumar S. Physiological functions of the HECT family of ubiquitin ligases. Nat Rev Mol Cell Biol. 2009; 10: 398-409. Ref.: https://goo.gl/eGyxs1
- Smalle J, Vierstra RD. The ubiquitin 26S proteasome proteolytic pathway. Annu Rev Plant Biol. 2004; 55: 555-590. Ref.: https://goo.gl/VNxcZu
- Vierstra RD. The ubiquitin/26S proteasome pathway, the complex last chapter in the life of many plant proteins. Trends Plant Sci. 2003; 8: 135-142. Ref.: https://goo.gl/vh46mo
- Glickman MH, Ciechanover A. The ubiquitin-proteasome proteolytic pathway: destruction for the sake of construction. Physiol Rev. 2002; 82: 373-428. Ref.: https://goo.gl/bsGuJv
- Hershko A, Ciechanover A. The ubiquitin system. Annu Rev Biochem. 1998; 67: 425-479. Ref.: https://goo.gl/Fb5bzy
- Scheffner M, Nuber U, Huibregtse JM. Protein ubiquitination involving an E1-E2-E3 enzyme ubiquitin thioester cascade. Nature. 1995; 373: 81-83. Ref.: https://goo.gl/7Zxck4
- Wang M, Cheng D, Peng J, Pickart CM. Molecular determinants of polyubiquitin linkage selection by an HECT ubiquitin ligase. EMBO J. 2006; 25: 1710-1719. Ref.: https://goo.gl/5bpDRk
- Peng J, Schwartz D, Elias JE, Thoreen CC, Cheng D, et al. A proteomics approach to understanding protein ubiquitination. Nat Biotechnol. 2003; 21: 921-926. Ref.: https://goo.gl/8reR9z
- Kulathu Y, Komander D. Atypical ubiquitylation - the unexplored world of polyubiquitin beyond Lys48 and Lys63 linkages. Nat Rev Mol Cell Biol. 2012; 13: 508-523. Ref.: https://goo.gl/MGUz25
- Mukhopadhyay D, Riezman H. Proteasome-independent functions of ubiquitin in endocytosis and signaling. Science. 2007; 315: 201-205. Ref.: https://goo.gl/GuWbds
- Thrower JS, Hoffman L, Rechsteiner M, Pickart CM. Recognition of the polyubiquitin proteolytic signal. EMBO J. 2000; 19: 94-102. Ref.: https://goo.gl/L1bFNc
- Xu P, Duong DM, Seyfried NT, Cheng D, Xie Y, et al. Quantitative proteomics reveals the function of unconventional ubiquitin chains in proteasomal degradation. Cell. 2009; 137: 133-145. Ref.: https://goo.gl/rCkV7b
- Metzger MB, Hristova VA, Weissman AM. HECT and RING finger families of E3 ubiquitin ligases at a glance. J Cell Sci. 2012; 125: 531-537. Ref.: https://goo.gl/AjcYM8
- Varshavsky A. The ubiquitin system, an immense realm. Annu Rev Biochem.2012; 81: 167-176. Ref.: https://goo.gl/Kj5VeD
- Schwartz AL, Ciechanover A. Targeting proteins for destruction by the ubiquitin system: implications for human pathobiology. Annu Rev Pharmacol Toxicol.2009; 49: 73-96. Ref.: https://goo.gl/8U8nG2
- Mazzucotelli E, Belloni S, Marone D, De Leonardis A, Guerra D, et al. The e3 ubiquitin ligase gene family in plants: regulation by degradation. Curr Genomics. 2006; 7: 509-522. Ref.: https://goo.gl/gvgc9c
- Chen L, Hellmann H. Plant E3 ligases: flexible enzymes in a sessile world. Mol Plant. 2013; 6: 1388-1404. Ref.: https://goo.gl/BSNDzM
- Hotton SK, Callis J. Regulation of cullin RING ligases. Annu Rev Plant Biol.2008; 59: 467-489. Ref.: https://goo.gl/1fcvky
- Schwechheimer C, Calderon Villalobos LI. Cullin-containing E3 ubiquitin ligases in plant development. Curr Opin Plant Biol. 2004; 7: 677-686. Ref.: https://goo.gl/2UueSJ
- Smalle J, Vierstra RD. The ubiquitin 26S proteasome proteolytic pathway. Annu Rev Plant Biol.2004; 55: 555-590. Ref.: https://goo.gl/gfTEC4
- Li W, Bengtson MH, Ulbrich A, Matsuda A, Reddy VA, et al. Genome-wide and functional annotation of human E3 ubiquitin ligases identifies MULAN, a mitochondrial E3 that regulates the organelle's dynamics and signaling. PloS One. 2008; 3: e1487. Ref.: https://goo.gl/zTqDRL
- Scheffner M, Staub O. HECT E3s and human disease. BMC Biochemistry. 2007; 8: S6. Ref.: https://goo.gl/6uucSJ
- Wang D, Ma L, Wang B, Liu J, Wei W. E3 ubiquitin ligases in cancer and implications for therapies. Cancer Metastasis Rev. 2017; 36: 683-702. Ref.: https://goo.gl/yPLu6T
- Lee JH, Kim WT. Regulation of abiotic stress signal transduction by E3 ubiquitin ligases in Arabidopsis. Mol Cells.2011; 31: 201-208. Ref.: https://goo.gl/Ta4tBZ
- Marino D, Froidure S, Canonne J, Ben Khaled S, Khafif M, et al. Arabidopsis ubiquitin ligase MIEL1 mediates degradation of the transcription factor MYB30 weakening plant defence. Nat Commun. 2013; 4: 1476. Ref.: https://goo.gl/4SBZw5
- Yee D, Goring DR. The diversity of plant U-box E3 ubiquitin ligases: from upstream activators to downstream target substrates. J Exp Bot. 2009; 60: 1109-1121. Ref.: https://goo.gl/FcRFRg
- Marin I. Evolution of plant HECT ubiquitin ligases. PloS one. 2013; 8: 68536. Ref.: https://goo.gl/Zv4Sn7
- https://goo.gl/1vfTxG
- Mund T, Lewis MJ, Maslen S, Pelham HR. Peptide and small molecule inhibitors of HECT-type ubiquitin ligases. Proceedings of the National Academy of Sciences of the United States of America. 2014; 111: 16736-16741. Ref.: https://goo.gl/vonLFC
- Opperman KJ, Mulcahy B, Giles AC, Risley MG, Birnbaum RL, et al. The HECT Family Ubiquitin Ligase EEL-1 Regulates Neuronal Function and Development. Cell reports. 2017; 19: 822-835. Ref.: https://goo.gl/jCVGid
- Kim HC, Steffen AM, Oldham ML, Chen J, Huibregtse JM. Structure and function of a HECT domain ubiquitin-binding site. EMBO reports. 2011; 12: 334-341. Ref.: https://goo.gl/zWr4Eo
- Maspero E, Valentini E, Mari S, Cecatiello V, Soffientini P, et al. Structure of a ubiquitin-loaded HECT ligase reveals the molecular basis for catalytic priming. Nature structural & molecular biology. 2013; 20: 696-701. Ref.: https://goo.gl/sgae9H
- Downes BP, Stupar RM, Gingerich DJ, Vierstra RD. The HECT ubiquitin-protein ligase (UPL) family in Arabidopsis: UPL3 has a specific role in trichome development. The Plant journal : for cell and molecular biology. 2003; 35: 729-742. Ref.: https://goo.gl/vS6oj8
- Verdecia MA, Joazeiro CA, Wells NJ, Ferrer JL, Bowman ME, et al. Conformational flexibility underlies ubiquitin ligation mediated by the WWP1 HECT domain E3 ligase. Molecular cell. 2003; 11: 249-259. Ref.: https://goo.gl/5zUPkq
- Huang L, Kinnucan E, Wang G, Beaudenon S, Howley PM, et al. Structure of an E6AP-UbcH7 complex: insights into ubiquitination by the E2-E3 enzyme cascade. Science. 1999; 286: 1321-1326. Ref.: https://goo.gl/ncU7C7
- Grau-Bove X, Sebe-Pedros A, Ruiz-Trillo I. A genomic survey of HECT ubiquitin ligases in eukaryotes reveals independent expansions of the HECT system in several lineages. Genome biology and evolution. 2013; 5: 833-847. Ref.: https://goo.gl/z1um7e
- Altschul SF, Madden TL, Schaffer AA, Zhang J, Zhang Z, Miller W, et al. Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic acids research. 1997; 25: 3389-3402. Ref.: https://goo.gl/Pa9As4
- Kumar S, Stecher G, Tamura K. MEGA7: Molecular Evolutionary Genetics Analysis Version 7.0 for Bigger Datasets. Molecular biology and evolution. 2016; 33: 1870-1874. Ref.: https://goo.gl/gk7sRb
- Hu B, Jin J, Guo AY, Zhang H, Luo J, Gao G. GSDS 2.0: an upgraded gene feature visualization server. Bioinformatics (Oxford, England). 2015; 31: 1296-1297. Ref.: https://goo.gl/KpnLFf
- Finn RD, Bateman A, Clements J, Coggill P, Eberhardt RY, et al. Pfam: the protein families database. Nucleic acids research. 2014; 42: 222-230.
- Mitchell A, Chang HY, Daugherty L, Fraser M, Hunter S, et al. The InterPro protein families database: the classification resource after 15 years. Nucleic acids research. 2015; 43: 213-221. Ref.: https://goo.gl/VCqgcq
- Hofmann K, Bucher P. The UBA domain: a sequence motif present in multiple enzyme classes of the ubiquitination pathway. Trends in biochemical sciences. 1996; 21: 172-173. Ref.: https://goo.gl/yk8qo9
- Hofmann K, Falquet L. A ubiquitin-interacting motif conserved in components of the proteasomal and lysosomal protein degradation systems. Trends in biochemical sciences. 2001; 26: 347-350. Ref.: https://goo.gl/GDPMaQ
- El Refy A, Perazza D, Zekraoui L, Valay JG, Bechtold N, et al. The Arabidopsis KAKTUS gene encodes a HECT protein and controls the number of endoreduplication cycles. Molecular genetics and genomics: MGG. 2003; 270: 403-414. Ref.: https://goo.gl/NGUBT6
- Bates PW, Vierstra RD. UPL1 and 2, two 405 kDa ubiquitin-protein ligases from Arabidopsis thaliana related to the HECT-domain protein family. The Plant journal: for cell and molecular biology. 1999; 20: 183-195. Ref.: https://goo.gl/UhEDbe
- Wang M, Pickart CM. Different HECT domain ubiquitin ligases employ distinct mechanisms of polyubiquitin chain synthesis. The EMBO journal. 2005; 24: 4324-4333. Ref.: https://goo.gl/6xD8Lm
- Galan JM, Haguenauer-Tsapis R. Ubiquitin lys63 is involved in ubiquitination of a yeast plasma membrane protein. The EMBO journal. 1997; 16: 5847-5854. Ref.: https://goo.gl/49euRF
- Chau V, Tobias JW, Bachmair A, Marriott D, Ecker DJ, Gonda DK, et al. A multiubiquitin chain is confined to specific lysine in a targeted short-lived protein. Science. 1989; 243: 1576-1583. Ref.: https://goo.gl/XgCpRj
- Mani A, Oh AS, Bowden ET, Lahusen T, Lorick KL, et al. E6AP mediates regulated proteasomal degradation of the nuclear receptor coactivator amplified in breast cancer 1 in immortalized cells. Cancer research. 2006; 66: 8680-8686. Ref.: https://goo.gl/V7JLQ4
- Finley D, Sadis S, Monia BP, Boucher P, Ecker DJ, et al. Inhibition of proteolysis and cell cycle progression in a multiubiquitination-deficient yeast mutant. Molecular and cellular biology. 1994; 14: 5501-5509. Ref.: https://goo.gl/rKPdVb
- Louria-Hayon I, Alsheich-Bartok O, Levav-Cohen Y, Silberman I, Berger M, et al. E6AP promotes the degradation of the PML tumor suppressor. Cell death and differentiation. 2009; 16: 1156-1166. Ref.: https://goo.gl/QXz1X3
- Adhikary S, Marinoni F, Hock A, Hulleman E, Popov N, et al. The ubiquitin ligase HectH9 regulates transcriptional activation by Myc and is essential for tumor cell proliferation. Cell. 2005; 123: 409-421. Ref.: https://goo.gl/JxTvYL
- Chen D, Kon N, Li M, Zhang W, Qin J, et al. ARF-BP1/Mule is a critical mediator of the ARF tumor suppressor. Cell. 2005; 121: 1071-1083. Ref.: https://goo.gl/6uRPyA
- Zhong Q, Gao W, Du F, Wang X. Mule/ARF-BP1, a BH3-only E3 ubiquitin ligase, catalyzes the polyubiquitination of Mcl-1 and regulates apoptosis. Cell. 2005; 121: 1085-1095. Ref.: https://goo.gl/Jujmb4
- Zhao X, Heng JI, Guardavaccaro D, Jiang R, Pagano M, et al. The HECT-domain ubiquitin ligase Huwe1 controls neural differentiation and proliferation by destabilizing the N-Myc oncoprotein. Nature cell biology. 2008; 10: 643-653. Ref.: https://goo.gl/gepC9k
- Patra B, Pattanaik S, Yuan L. Ubiquitin protein ligase 3 mediates the proteasomal degradation of GLABROUS 3 and ENHANCER OF GLABROUS 3, regulators of trichome development and flavonoid biosynthesis in Arabidopsis. The Plant journal : for cell and molecular biology. 2013; 74: 435-447. Ref.: https://goo.gl/R9pcC6
- Miao Y, Zentgraf U. A HECT E3 ubiquitin ligase negatively regulates Arabidopsis leaf senescence through degradation of the transcription factor WRKY53. The Plant journal: for cell and molecular biology. 2010; 63: 179-188. Ref.: https://goo.gl/h3RnAN
- Deng S, Jang IC, Su L, Xu J, Chua NH. JMJ24 targets CHROMOMETHYLASE3 for proteasomal degradation in Arabidopsis. Genes & development. 2016; 30: 251-256. Ref.: https://goo.gl/y8Ti6K
- Zeng S, Wang Y, Zhang T, Bai L, Wang Y, et al. E3 ligase UHRF2 stabilizes the acetyltransferase TIP60 and regulates H3K9ac and H3K14ac via RING finger domain. Protein & cell. 2017; 8: 202-218. Ref.: https://goo.gl/cXXwDZ
- Luo C, Cai X T, Du J, Zhao TL, Wang PF, et al. PARAQUAT TOLERANCE3 Is an E3 Ligase That Switches off Activated Oxidative Response by Targeting Histone-Modifying PROTEIN METHYLTRANSFERASE4b. PLoS genetics. 2016; 12: 1006332. Ref.: https://goo.gl/MSw78F
- Licchesi JD, Mieszczanek J, Mevissen TE, Rutherford TJ, Akutsu M, et al. An ankyrin-repeat ubiquitin-binding domain determines TRABID's specificity for atypical ubiquitin chains. Nature structural & molecular biology. 2011; 19: 62-71. Ref.: https://goo.gl/ms7xPM
- Michel MA, Elliott PR, Swatek KN, Simicek M, Pruneda JN, et al. Assembly and specific recognition of k29- and k33-linked polyubiquitin. Molecular cell. 2015; 58: 95-109. Ref.: https://goo.gl/9rhBzm
- Jin J, Xie X, Xiao Y, Hu H, Zou Q, et al. Epigenetic regulation of the expression of Il12 and Il23 and autoimmune inflammation by the deubiquitinase Trabid. Nature immunology. 2016; 17: 259-268. Ref.: https://goo.gl/KonC54
- Huibregtse JM, Yang JC, Beaudenon SL. The large subunit of RNA polymerase II is a substrate of the Rsp5 ubiquitin-protein ligase. Proceedings of the National Academy of Sciences of the United States of America. 1997; 94: 3656-3661. Ref.: https://goo.gl/as7SRp
- Somesh BP, Sigurdsson S, Saeki H, Erdjument-Bromage H, Tempst P, et al. Communication between distant sites in RNA polymerase II through ubiquitylation factors and the polymerase CTD. Cell. 2007; 129: 57-68. Ref.: https://goo.gl/epiDCi
- Gao M, Labuda T, Xia Y, Gallagher E, Fang D, Liu YC, et al. Jun turnover is controlled through JNK-dependent phosphorylation of the E3 ligase Itch. Science. 2004; 306: 271-275. Ref.: https://goo.gl/i32gz8
- JC C. Armadillo repeat proteins: versatile regulators of plant development and signalling. Plant Cell Monographs. 2008; 10: 299-314. Ref.: https://goo.gl/4g68Wj
- Lan W, Qiu S, Ren Y, Miao Y. Expression Profile and Function of HECT E3s in Arabidopsis thaliana. Acta Botanica Boreali-Occidentalia Sinica. 2017; 37: 2112-2119.
- Fu H, Sadis S, Rubin DM, Glickman M, van Nocker S, Finley D, et al. Multiubiquitin chain binding and protein degradation are mediated by distinct domains within the 26 S proteasome subunit Mcb1. The Journal of biological chemistry. 1998; 273: 1970-1981. Ref.: https://goo.gl/3Wj5Ab
- Sharma M, Singh A, Shankar A, Pandey A, Baranwal V, et al. Comprehensive expression analysis of rice Armadillo gene family during abiotic stress and development. DNA research: an international journal for rapid publication of reports on genes and genomes. 2014; 21: 267-283. Ref.: https://goo.gl/tGkaqh
- Wang H, Lu Y, Jiang T, Berg H, Li C, et al. The Arabidopsis U-box/ARM repeat E3 ligase AtPUB4 influences growth and degeneration of tapetal cells, and its mutation leads to conditional male sterility. Plant J. 2013; 74: 511-523. Ref.: https://goo.gl/nq6h3W
- Winter D, Vinegar B, Nahal H, Ammar R, Wilson GV, et al. An "Electronic Fluorescent Pictograph" browser for exploring and analyzing large-scale biological data sets. PloS one. 2007; 2. Ref.: https://goo.gl/aHB14q
- M H. Ubiquitin-dependent protein degradation. Annu Rev Genet. 1996; 30: 405-439.
Figures:
Figure 1
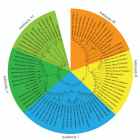
Figure 2

Figure 3
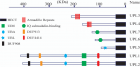
Figure 4

Figure 5
Similar Articles
-
Role of HECT ubiquitin protein ligases in Arabidopsis thalianaYing Miao*,Wei Lan,Weibo Ma. Role of HECT ubiquitin protein ligases in Arabidopsis thaliana. . 2018 doi: 10.29328/journal.jpsp.1001016; 2: 020-030
-
Evaluation of cold response in Ilex paraguariensisCristian Antonio Rojas*,Sandonaid Andrei Geisler,Carina Francisca Argüelles. Evaluation of cold response in Ilex paraguariensis. . 2019 doi: 10.29328/journal.jpsp.1001026; 3: 009-012
-
Cloning and Characterization of a Pseudo-Response Regulator 7 (PRR7) Gene from Medicago Sativa Involved In Regulating the Circadian ClockWeifeng Nian*,Yilin Shen. Cloning and Characterization of a Pseudo-Response Regulator 7 (PRR7) Gene from Medicago Sativa Involved In Regulating the Circadian Clock. . 2019 doi: 10.29328/journal.jpsp.1001032; 3: 056-061
-
MALDI-MSI method for the detection of large biomolecules in plant leaf tissueLilian ST Carmo,Daiane G Ribeiro,Eder A Barbosa,Luciano P Silva,Angela Mehta*. MALDI-MSI method for the detection of large biomolecules in plant leaf tissue. . 2021 doi: 10.29328/journal.jpsp.1001061; 5: 058-061
Recently Viewed
-
Knowledge, Attitude, and Practice of Healthcare Workers in Ekiti State, Nigeria on Prevention of Cervical CancerAde-Ojo Idowu Pius*, Okunola Temitope Omoladun, Olaogun Dominic Oluwole. Knowledge, Attitude, and Practice of Healthcare Workers in Ekiti State, Nigeria on Prevention of Cervical Cancer. Arch Cancer Sci Ther. 2024: doi: 10.29328/journal.acst.1001038; 8: 001-006
-
Oral Cancer Management is not just Treatment! But also, how early Pre-cancerous Lesions are Diagnosed & Treated!!Suresh Kishanrao*. Oral Cancer Management is not just Treatment! But also, how early Pre-cancerous Lesions are Diagnosed & Treated!!. Arch Cancer Sci Ther. 2024: doi: 10.29328/journal.acst.1001039; 8: 007-012
-
Breast Cancer in FemaleLorena Menditto*. Breast Cancer in Female. Arch Cancer Sci Ther. 2024: doi: 10.29328/journal.acst.1001040; 8: 013-018
-
Lived Experiences of Cervical Cancer Patients Receiving Chemotherapy at Cancer Diseases Hospital in Lusaka, ZambiaElisha Benkeni Kapya*, Marjorie Kabinga Makukula, Mwaba Chileshe Siwale, Victoria Kalusopa Mwiinga, Elijah Mpundu. Lived Experiences of Cervical Cancer Patients Receiving Chemotherapy at Cancer Diseases Hospital in Lusaka, Zambia. Arch Cancer Sci Ther. 2024: doi: 10.29328/journal.acst.1001041; 8: 019-036
-
Accessory Splenic Mass Masquerading as Hepatocellular Carcinoma: A Diagnostic DilemmaSoe P Winn*, Tharun Shyam, M Isabel Fiel, Yiwu Huang. Accessory Splenic Mass Masquerading as Hepatocellular Carcinoma: A Diagnostic Dilemma. Arch Cancer Sci Ther. 2024: doi: 10.29328/journal.acst.1001042; 8: 037-040
Most Viewed
-
Evaluation of Biostimulants Based on Recovered Protein Hydrolysates from Animal By-products as Plant Growth EnhancersH Pérez-Aguilar*, M Lacruz-Asaro, F Arán-Ais. Evaluation of Biostimulants Based on Recovered Protein Hydrolysates from Animal By-products as Plant Growth Enhancers. J Plant Sci Phytopathol. 2023 doi: 10.29328/journal.jpsp.1001104; 7: 042-047
-
Sinonasal Myxoma Extending into the Orbit in a 4-Year Old: A Case PresentationJulian A Purrinos*, Ramzi Younis. Sinonasal Myxoma Extending into the Orbit in a 4-Year Old: A Case Presentation. Arch Case Rep. 2024 doi: 10.29328/journal.acr.1001099; 8: 075-077
-
Feasibility study of magnetic sensing for detecting single-neuron action potentialsDenis Tonini,Kai Wu,Renata Saha,Jian-Ping Wang*. Feasibility study of magnetic sensing for detecting single-neuron action potentials. Ann Biomed Sci Eng. 2022 doi: 10.29328/journal.abse.1001018; 6: 019-029
-
Pediatric Dysgerminoma: Unveiling a Rare Ovarian TumorFaten Limaiem*, Khalil Saffar, Ahmed Halouani. Pediatric Dysgerminoma: Unveiling a Rare Ovarian Tumor. Arch Case Rep. 2024 doi: 10.29328/journal.acr.1001087; 8: 010-013
-
Physical activity can change the physiological and psychological circumstances during COVID-19 pandemic: A narrative reviewKhashayar Maroufi*. Physical activity can change the physiological and psychological circumstances during COVID-19 pandemic: A narrative review. J Sports Med Ther. 2021 doi: 10.29328/journal.jsmt.1001051; 6: 001-007

HSPI: We're glad you're here. Please click "create a new Query" if you are a new visitor to our website and need further information from us.
If you are already a member of our network and need to keep track of any developments regarding a question you have already submitted, click "take me to my Query."