Abstract
Research Article
Cold Atmospheric Pressure Plasma Jet and Plasma Lamp Interaction with Plants: Electrostimulation, Reactive Oxygen and Nitrogen Species, and Side Effects
Alexander G Volkov*, Jewel S Hairston, Darayas Patel and Sergey Sarkisov
Published: 22 August, 2023 | Volume 7 - Issue 2 | Pages: 081-088
Cold atmospheric pressure plasma (CAPP) treatment is a highly effective method of protecting seeds, plants, flowers, and trees from diseases and infection and significantly increasing crop yields.
Here we found that cold atmospheric pressure He-plasma jet (CAPPJ) can also cause side effects and damage to plants if the plasma exposure time is too long. Reactive oxygen and nitrogen species (RONS), electromagnetic fields, and ultraviolet photons emitted by CAPPJ can cause both positive and negative effects on plants. CAPPJ can interact with biological tissue surfaces. The plasma lamp has no visible side effects on Aloe vera plants, cabbage, and tomatoes. A plasma lamp and a cold atmospheric pressure plasma He-jet cause strong electrical signaling in plants with a very high amplitude with frequencies equal to the frequency of plasma generation. The use of plasma lamps for electrostimulation of biological tissues can help to avoid side processes in biological tissues associated with the generation of RONS, UV photons, and direct interaction with cold plasma. CAPP technology can play an important role in agriculture, medicine, the food industry, chemistry, surface science, material science, and engineering applications without side effects if the plasma exposure is short enough.
Read Full Article HTML DOI: 10.29328/journal.jpsp.1001110 Cite this Article Read Full Article PDF
Keywords:
Cold plasma; Phytopathology; Plasma phytoelectrophysiology; Reactive oxygen and nitrogen species; Plant signaling; Plasma agriculture
References
- Dubinov AE, Lazarenko ER, Selemir VD. Effect of glow discharge air plasma on grain crops seed. IEEE Trans Plasma Sci. 2000; 28:180-183. doi 10.1109/27.842898.
- Filipov AK, Fedorov MA, Filipov DA. Method of pre-sowing treatment of plant seeds. Russian patent RU2293456C1, 2007.
- Yan D, Lin L, Zvansky M, Kohanzadeh L, Taban S, Chriqui S, Keidar M. Improving Seed Germination by cold atmospheric plasma. Plasma. 2022; 5:98-110. https://doi.org/10.3390/plasma5010008.
- Volkov AG, Hairston JS, Marshal J, Bookal A, Dholichand A, Patel D. Plasma seeds: Cold plasma accelerates Phaseolus vulgaris seeds' imbibition, germination, and speed of seedling growth. Plasma Med. 2020; 10:139–158. https:/doi.org/10.1615/PlasmaMed.2020036438.
- Volkov AG, Hairston JS, Patel D, Gott RK, Xu KG. Cold plasma poration and corrugation of pumpkin seed coats. Bioelectrochem. 2019; 128:175-185. doi:10.1016/j.bioelechem.2019.04.012.
- Volkov AG, Xu KG, Kolobov VI. Cold plasma interactions with plants: Morphing and movements of Venus flytrap and Mimosa pudica induced by argon plasma jet. Bioelectrochemistry. 2017 Dec;118:100-105. doi: 10.1016/j.bioelechem.2017.07.011. Epub 2017 Jul 29. PMID: 28780442.
- Volkov AG, Xu KG, Kolobov VI. Plasma-generated reactive oxygen and nitrogen species can lead to closure, locking and constriction of the Dionaea muscipula Ellis trap. J R Soc Interface. 2019 Jan 31;16(150):20180713. doi: 10.1098/rsif.2018.0713. PMID: 30958146; PMCID: PMC6364641.
- Volkov AG. Cold atmospheric pressure He-plasma jet and plasma ball interactions with the Venus flytrap: Electrophysiology and side effects. Bioelectrochemistry. 2021 Aug;140:107833. doi: 10.1016/j.bioelechem.2021.107833. Epub 2021 May 1. PMID: 33989989.
- Nakano R, Tashiro K, Aijima R, Hayashi N. Effect of oxygen plasma irradiation on gene expression in plant seeds induced by active oxygen species. Plasma Med. 2016; 6:303-13.
- Pua N, Zivkovi S, Selakovi N, Milutinovi M, Boljevi J, Malovi G, Petrovi Z. Long and short-term effects of plasma treatment on meristematic plant cells. Appl Phys Lett. 2014; 104:214106. doi 10.1063/1.4880360.
- Kobayashi M, Wang Y, Kumagai S, Uraoka Y, Ito Effects of cold atmospheric plasma irradiation on Arabidopsis seedlings. Japanese J Appl Physics. 2020; 59: SAAB09. doi:10.7567/1347-4065/ab4e7b.
- Lazović S, Maletić D, Leskovac A, Filipović J, Puač N, Malović G, Joksić G, Petrović Z. Plasma induced DNA damage: comparison with the effects of ionizing radiation. Appl Phys Lett. 2014; 105:124101. doi: 10.1063/1.4896626
- Adhikari B, Adhikari M, Ghimire B, Adhikari BC, Park G, Choi EH. Cold plasma seed priming modulates growth, redox homeostasis and stress response by inducing reactive species in tomato (Solanum lycopersicum). Free Radic Biol Med. 2020 Aug 20;156:57-69. doi: 10.1016/j.freeradbiomed.2020.06.003. Epub 2020 Jun 17. PMID: 32561321.
- Tomeková J, Kyzek S, Medvecká V, Gálová E, Zahoranová Influence of cold atmospheric pressure plasma on pea seeds: DNA damage of seedlings and optical diagnostics of plasma. Plasma Chem. Plasma Proces. 2020; 40:1571–1584.
- Marzban M, Farahani F, Atyabi SM, Noormohammadi Z. Induced genetic and chemical changes in medicinally important plant Catharanthus roseus (L.) G. Don: cold plasma and phytohormones. Mol Biol Rep. 2022 Jan;49(1):31-38. doi: 10.1007/s11033-021-06789-w. Epub 2021 Nov 13. PMID: 34773551.
- Seol YB, Kim J, Park SH, Young Chang H. Atmospheric Pressure Pulsed Plasma Induces Cell Death in Photosynthetic Organs via Intracellularly Generated ROS. Sci Rep. 2017 Apr 3;7(1):589. doi: 10.1038/s41598-017-00480-6. PMID: 28373681; PMCID: PMC5428426.
- Švubová R, Kyzek S, Medvecká V, Slováková I, Gálová E, Zahoranová Novel insight at the effect of cold atmospheric pressure plasma on the activity of enzymes essential for the germination of pea (Pisum sativum L. cv. Prophet) Seeds. Plasma Chem. Plasma Processing. 2020; 40:1221–40.
- Tesla Incandescent electric light. U.S. Patent Number 0.514.170, 1894.
- Burin MJ, Simons GG, Ceja HG, Zweben SJ, Nagy A, Brunkhorst On filament structure and propagation within a commercial plasma globe. Phys Plasmas. 2015; 22:053509. doi:10.1063/1.4919939.
- Volkov AG, Bookal A, Hairston JS, Patel D. Radio frequency plasma capacitor can increase rates of seeds imbibition, germination, and radicle growth. Funct Plant Biol. 2021 Feb;48(3):312-320. doi: 10.1071/FP20293. PMID: 33220717.
- Ark A, Parry Application of high-frequency electrostatic fields in agriculture. The Quaternary Review Biol. 1940; 15:172-91.
- Bertholon MDE. The electricity of plants: Book in which we deal with the electricity of the atmosphere on plants, its effects on the economy of plants, their medical virtues. (P.F. Didotjeune, Paris, France) 1783.
- Bois-Reymond DUE. Investigations on animal electricity. 1: 7-10. (Published by G. Reimer, Berlin, Germany) 1848.
- Bose J Transmission of Stimuli in Plants. Nature. 1925; 115:457-7.
- Ksenzhek OS; Volkov A Plant Energetics. (Academic Press, San Diego, US) 1998.
- Volkov A Plant Electrophysiology. Methods and Cell Electrophysiology. (Springer, Berlin, Germany) 2012.
- Volkov A Plant Electrophysiology. Signaling and Responses. (Springer, Berlin, Germany) 2012.
- Solly On the influence of electricity on vegetation. J Horticultural Soc London. 1846; 1:81-109.
- Murr L Plant growth response in a simulated electric field environment. Nature. 1963; 200:490–491. doi 10.1038/200490b0.
- Lemström Electricity in Agriculture and Horticulture. (Electrician Publications, London, UK, 1904.
- Volkov AG, Foster JC, Jovanov E, Markin VS. Anisotropy and nonlinear properties of electrochemical circuits in leaves of Aloe vera L. Bioelectrochemistry. 2011 Apr;81(1):4-9. doi: 10.1016/j.bioelechem.2010.11.001. Epub 2010 Nov 27. PMID: 21167797.
- Volkov AG, O’Neal L, Volkova-Gugeshashvili MI, Markin V Electrostimulation of Aloe Vera L., Mimosa Pudica L. and Arabidopsis Thaliana: Propagation and collision of electronic potentials. J Electrochem Soc. 2013; 160:G3102-G3111.
- Volkov AG, Shtessel YB. Electrical signal propagation within and between tomato plants. Bioelectrochemistry. 2018 Dec;124:195-205. doi: 10.1016/j.bioelechem.2018.08.001. Epub 2018 Aug 7. PMID: 30125795.
- Jack JJ, Noble D, Tsien R Electric Current Flow in Excitable Cells. Clarendon, Oxford, 1975.
- Hodgkin AL, Rushton WA. The electrical constants of a crustacean nerve fibre. Proc R Soc Med. 1946 Dec 3;134(873):444-79. doi: 10.1098/rspb.1946.0024. PMID: 20281590.
- Rall Time constants and electrotonic length of membrane cylinders and neurons. Biophys J. 1969; 58:631-639.
- Kyzek S, Holubova L, Medvecká V, Tomekova J, Gálová E, Zahoranová Cold atmospheric pressure plasma can induce an adaptive response in pea seeds. Plasma Chem. Plasma Processing. 2019; doi: 10.1007s11090-018-9951-x.
- Stolarik T, Henselova M, Martinka M, Novak O, Zahoranova A, Cernak Effect of low-temperature plasma on the structure of seeds, growth, and metabolism of exogenous phytohormones in pea (Pisum sativum L.) Plasma Chemistry and Plasma Processing. 2015. doi: 10.1007/s11090-015-9627-8
- Lackmann JW, Schneider S, Edengeiser E, Jarzina F, Brinckmann S, Steinborn E, Havenith M, Benedikt J, Bandow JE. Photons and particles emitted from cold atmospheric-pressure plasma inactivate bacteria and biomolecules independently and synergistically. J R Soc Interface. 2013 Sep 25;10(89):20130591. doi: 10.1098/rsif.2013.0591. PMID: 24068175; PMCID: PMC3808546.
- Lv X, Cheng JH. Evaluation of the Effects of Cold Plasma on Cell Membrane Lipids and Oxidative Injury of Salmonella typhimurium. Molecules. 2022 Jan 19;27(3):640. doi: 10.3390/molecules27030640. PMID: 35163904; PMCID: PMC8838372.
- Attri P, Kumar N, Park JH, Yadav DK, Choi S, Uhm HS, Kim IT, Choi EH, Lee W. Influence of reactive species on the modification of biomolecules generated from the soft plasma. Sci Rep. 2015 Feb 4;5:8221. doi: 10.1038/srep08221. PMID: 25649786; PMCID: PMC4316168.
- Takai E, Kitamura T, Kuwabara J, Ikawa S, Yoshizawa S, Shiraki K, Kawasaki H, Arakawa R, Kitano Chemical modification of amino acids by atmospheric-pressure cold plasma in aqueous solution. J Phys D Appl Phys. 2014; 47:285403. https://doi.org/10.1088/0022-3727/47/28/285403.
- Van Breusegem F, Dat JF. Reactive oxygen species in plant cell death. Plant Physiol. 2006 Jun;141(2):384-90. doi: 10.1104/pp.106.078295. PMID: 16760492; PMCID: PMC1475453.
- Lu X, Naidis GV, Laroussi M, Reuter S, Graves DB, Ostrikov Reactive species in non-equilibrium atmospheric pressure plasmas: Generation, transport, and biological effects. Phys Rep. 2016; 630:1–84.
- Hill AC, Pack MR, Treshow M, Downs RJ, Transtrum L Plant injury induced by ozone. Phytopathology. 1961; 51:356-63.
- Patton RL, Garraway M Ozone-induced necrosis and increased peroxidase activity in hybrid poplar leaves. Environm. Experimental Bot. 1986; 26:137-141.
- Volkov AG, Bookal A, Hairston JS, Roberts J, Taengwa G, Patel Mechanisms of multielectron reactions at the plasma/water interface: Interfacial catalysis, RONS, nitrogen fixation, and plasma-activated water. Electrochim Acta. 2021; 385:138441. doi:10.1016/j.electacta.2021.138441.
- Volkov AG, Hairston JS, Taengwa G, Roberts J, Liburd L, Patel D. Redox Reactions of Biologically Active Molecules upon Cold Atmospheric Pressure Plasma Treatment of Aqueous Solutions. Molecules. 2022 Oct 19;27(20):7051. doi: 10.3390/molecules27207051. PMID: 36296644; PMCID: PMC9608965
Figures:

Figure 1

Figure 2

Figure 3
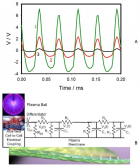
Figure 4
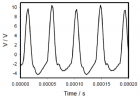
Figure 5
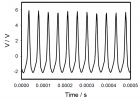
Figure 6

Figure 7
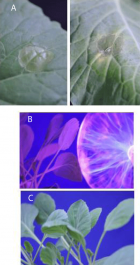
Figure 8
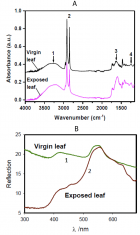
Figure 9
Figure 10
Similar Articles
-
Cold Atmospheric Pressure Plasma Jet and Plasma Lamp Interaction with Plants: Electrostimulation, Reactive Oxygen and Nitrogen Species, and Side EffectsAlexander G Volkov*, Jewel S Hairston, Darayas Patel, Sergey Sarkisov. Cold Atmospheric Pressure Plasma Jet and Plasma Lamp Interaction with Plants: Electrostimulation, Reactive Oxygen and Nitrogen Species, and Side Effects. . 2023 doi: 10.29328/journal.jpsp.1001110; 7: 081-088
Recently Viewed
-
Reliability and Diagnostic Performance of Transient Hepatic Elastography in Chronic Hepatitis C during the Training PhaseAppel F*, Ercolin S, Monteiro MM, Lomes N, Uehara S, Emori CT, Nunes EJS, El Bacha I, Oliveira AC, Feldner ACCA, Silva ISS, Silva AEB, Ferraz MLG, Parise ER, Carvalho-Filho RJ. Reliability and Diagnostic Performance of Transient Hepatic Elastography in Chronic Hepatitis C during the Training Phase. Ann Clin Gastroenterol Hepatol. 2024: doi: 10.29328/journal.acgh.1001047; 8: 021-027
-
Budesonide – Oral Galenic Formulations for Crohn DiseaseLuisetto M, Mashori GR, Cabianca L, Latyshev OYU. Budesonide – Oral Galenic Formulations for Crohn Disease. Ann Clin Gastroenterol Hepatol. 2024: doi: 10.29328/journal.acgh.1001048; 8: 028-033
-
Drug Rehabilitation Centre-based Survey on Drug Dependence in District Shimla Himachal PradeshKanishka Saini,Palak Sharma,Bhawna Sharma*,Atul Kumar Dubey,Muskan Bhatnoo,Prajkta Thakur,Vanshika Chandel,Ritika Sinha. Drug Rehabilitation Centre-based Survey on Drug Dependence in District Shimla Himachal Pradesh. J Addict Ther Res. 2025: doi: 10.29328/journal.jatr.1001032; 9: 001-006
-
A Rare Case of Uvulitis Following Endotracheal Intubation Using C- Mac Videolaryngoscope and How To Avoid ItKiran Kuruvilla George, Jini Chirackel Thomas, Krishnan B Sivaram, Krishna Prasad T*. A Rare Case of Uvulitis Following Endotracheal Intubation Using C- Mac Videolaryngoscope and How To Avoid It. Arch Pathol Clin Res. 2024: doi: 10.29328/journal.apcr.1001043; 8: 016-020
-
Gossypiboma due to a retained surgical sponge following abdominal hysterectomy, complicated by intestinal migration and small bowel obstruction- A Case ReportVivek Agrawal,Praroop Gupta*. Gossypiboma due to a retained surgical sponge following abdominal hysterectomy, complicated by intestinal migration and small bowel obstruction- A Case Report. Arch Surg Clin Res. 2018: doi: 10.29328/journal.ascr.1001017; 2: 015-017
Most Viewed
-
Evaluation of Biostimulants Based on Recovered Protein Hydrolysates from Animal By-products as Plant Growth EnhancersH Pérez-Aguilar*, M Lacruz-Asaro, F Arán-Ais. Evaluation of Biostimulants Based on Recovered Protein Hydrolysates from Animal By-products as Plant Growth Enhancers. J Plant Sci Phytopathol. 2023 doi: 10.29328/journal.jpsp.1001104; 7: 042-047
-
Sinonasal Myxoma Extending into the Orbit in a 4-Year Old: A Case PresentationJulian A Purrinos*, Ramzi Younis. Sinonasal Myxoma Extending into the Orbit in a 4-Year Old: A Case Presentation. Arch Case Rep. 2024 doi: 10.29328/journal.acr.1001099; 8: 075-077
-
Feasibility study of magnetic sensing for detecting single-neuron action potentialsDenis Tonini,Kai Wu,Renata Saha,Jian-Ping Wang*. Feasibility study of magnetic sensing for detecting single-neuron action potentials. Ann Biomed Sci Eng. 2022 doi: 10.29328/journal.abse.1001018; 6: 019-029
-
Pediatric Dysgerminoma: Unveiling a Rare Ovarian TumorFaten Limaiem*, Khalil Saffar, Ahmed Halouani. Pediatric Dysgerminoma: Unveiling a Rare Ovarian Tumor. Arch Case Rep. 2024 doi: 10.29328/journal.acr.1001087; 8: 010-013
-
Physical activity can change the physiological and psychological circumstances during COVID-19 pandemic: A narrative reviewKhashayar Maroufi*. Physical activity can change the physiological and psychological circumstances during COVID-19 pandemic: A narrative review. J Sports Med Ther. 2021 doi: 10.29328/journal.jsmt.1001051; 6: 001-007

HSPI: We're glad you're here. Please click "create a new Query" if you are a new visitor to our website and need further information from us.
If you are already a member of our network and need to keep track of any developments regarding a question you have already submitted, click "take me to my Query."